A Histotechnology Map of Drug Development Infographic
The Role of Histotechnology Across Drug Development
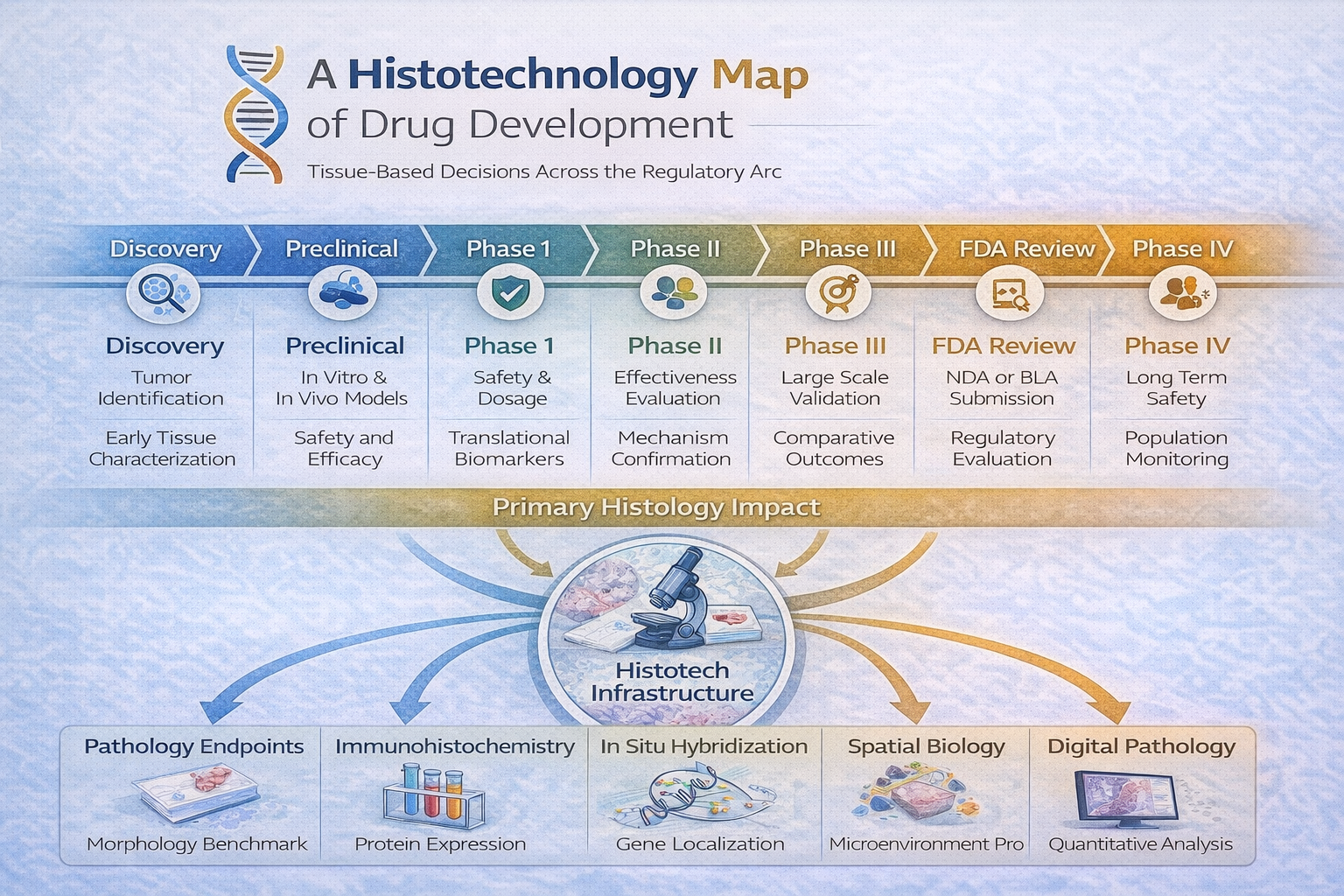
Discovery
Histotechnology supports early target validation through detailed tissue morphology, immunohistochemical localization, and spatial characterization of disease pathways. Tissue analysis helps confirm that proposed molecular targets are present and biologically relevant in situ.
Preclinical Research
In vitro and in vivo models rely on histology to assess toxicity, off-target effects, tumor architecture, and pharmacodynamic response. Xenograft and PDX models are validated through morphologic and species-specific markers to ensure model integrity.
Phase I
Histology contributes to translational biomarker alignment by evaluating human biopsy specimens for target engagement, apoptosis induction, proliferation changes, and early safety signals. Tissue findings are compared directly to preclinical benchmarks.
Phase II
Histologic endpoints support mechanism-of-action confirmation and dose–response relationships. Quantitative immunohistochemistry and image analysis help determine biologic activity across patient cohorts.
Phase III
Tissue-based biomarkers are used for large-scale validation of therapeutic effect, refinement of predictive markers, and correlation with clinical outcomes. Standardization and reproducibility across sites become critical.
FDA Review
Histologic data generated in earlier phases form part of the regulatory evidence package, supporting claims of safety, mechanism, and biomarker validity. Documentation integrity and assay validation are essential.
Phase IV (Post-Market Monitoring)
Although less routine, histotechnology may assist in investigating long-term safety signals, resistance mechanisms, or rare tissue-based adverse events identified after approval.