The Histotech Stack in Drug Development
Most people think histology is a technique.
In drug development, it is infrastructure.
In software engineering, a tech stack describes the layered technologies required to run a system. Databases, frameworks, servers. Each layer depends on the others.
Drug development works the same way.
Across oncology, toxicology, immunology, neuroscience, infectious disease, metabolic research, and regenerative medicine, histology functions as structured infrastructure.
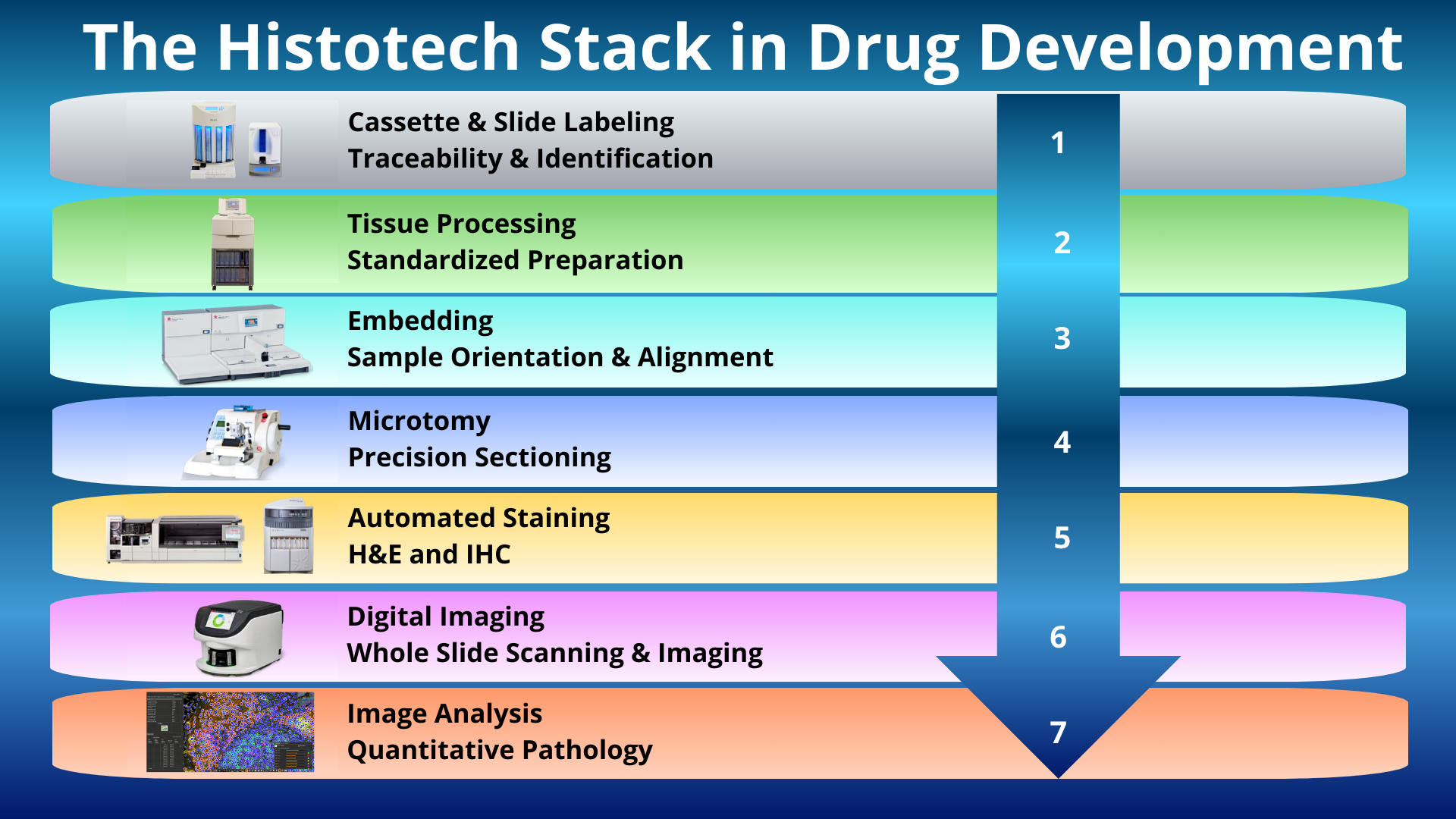
Let’s call it the Histotech Stack:
A layered histology technology infrastructure composed of integrated workflows, instrumentation, and analytic systems that collectively generate reproducible, tissue-based data in drug development.
What Is a Histotech Stack?
A Histotech Stack is the layered integrated structure of:
- Identification systems
- Processing platforms
- Embedding workflows
- Sectioning infrastructure
- Staining technologies
- Digital imaging systems
- Quantitative analysis platforms
A slide is not the result of skill alone.
It is the output of a system.
When one layer drifts, the interpretation drifts.
Visually, it looks like this:
Layer 1: Cassette and Slide Labeling
Studies across therapeutic areas involve:
- Multiple dose groups
- Timepoints
- Control arms
- Recovery cohorts
One labeling error compromises dataset integrity.
Traceability is not administrative. It is structural.
Instrumentation:
Layer 2: Tissue Processing
Processing defines what can be preserved and measured.
It affects:
- Morphologic stability
- Antigen preservation
- Nucleic acid integrity
- Special stain performance
Processing variability may not be immediately visible. It appears later as signal inconsistency, interpretive ambiguity, or quantification noise.
Instrumentation:
Layer 3: Embedding
Orientation determines:
- Margin and boundary representation
- Lesion distribution
- Tissue architecture preservation
- Spatial relationship between affected and normal regions
Embedding is alignment.
Misalignment alters interpretation across toxicology, oncology, inflammatory disease, and organ-specific studies.
Instrumentation:
Layer 4: Microtomy
Section thickness influences:
- Morphologic resolution
- Antigen and RNA accessibility
- Stain intensity
- Lesion detectability
- Quantitative reproducibility
Whether evaluating hepatotoxicity, neurodegeneration, fibrosis, immune infiltration, or tumor regression, variability introduced at the microtome propagates downstream.
Digital analysis assumes uniform input.
It does not correct mechanical drift.
Instrumentation:
- Leica CM1950 Cryostat
- Sakura Accu Cut SRM 300LT
- Microm HM 355
- Boekel Water Bath
- Biocare Desert Chamber Pro Oven
Layer 5: Automated Staining
Staining platforms support diverse endpoints:
- Routine morphology (H&E)
- Special stains for fibrosis, organisms, or structural change
- Immunohistochemistry for protein expression
- In situ hybridization for RNA localization
- Multiplex and spatial profiling methods
In drug development, staining supports:
- Safety evaluation
- Mechanism confirmation
- Biomarker validation
- Translational alignment
Consistency across runs and cohorts is essential for defensible interpretation.
Instrumentation:
- Tissue Tek Prizma Plus Automated Slide Stainer
- Tissue Tek Glas g2 Automated Glass Coverslipper
- Leica Bond RX and RXm
- Ventana Discovery Ultra
- Dako Autostainer Link 48
- Dako Omnis
Layer 6: Digital Imaging
Whole slide imaging transforms tissue sections into scalable data assets.
It enables:
- Remote review
- Archival stability
- Cross-site comparison
- Quantitative integration
The glass slide becomes part of a larger analytic system.
Instrumentation:
Layer 7: Image Analysis
Quantitative platforms convert morphology into measurable endpoints.
Examples include:
- Lesion area quantification
- Immune cell density
- Fibrosis scoring
- Proliferation and apoptosis indices
- Regional distribution mapping
Software does not create clarity.
It exposes variability introduced upstream.
Platforms:
Equipment Disclosure:
The instrumentation and platforms listed reflects the current laboratory stack at the time of publication and may change as platforms evolve. Vendor names are included for context only and do not constitute endorsement.
What the Stack Actually Protects
Histotechnologists think in sections.
Scientists think in endpoints.
Regulators think in documentation.
The stack connects all three.
When a dataset withstands scrutiny, it is because the infrastructure supported it.
When findings become ambiguous, the weakness is often upstream.
Not dramatic.
Incremental.
Processing variation.
Sectioning drift.
Staining inconsistency.
Calibration differences.
The Histotech Stack is not about producing slides.
It is about producing defensible data.
Its role is structural:
To convert tissue into reliable evidence across discovery, development, and regulation.